Prof. Arun Chougule PhD, FIUPESM, FIOMP, FAMS
Past President of AFOMP and Immediate past Chair of ETC IOMP
Vice President of Indian Society of Radiation Biology (ISRB)
arunchougule11@gmail.com
Introduction: From Physical Precision to Biological Precision
Over the last two decades, radiation oncology has witnessed remarkable technological advances. Techniques such as intensity-modulated radiotherapy (IMRT), stereotactic body radiotherapy (SBRT), and image-guided radiotherapy (IGRT) have allowed medical physicists to sculpt dose distributions with millimetric precision. Yet, despite similar dose–volume histograms (DVHs), clinicians frequently observe significant inter-patient variability in treatment response and normal tissue toxicity.
This inconsistency raises an important question: Are physical dose parameters alone sufficient to explain radiotherapy outcomes? Emerging evidence suggests that the gut microbiome, the complex community of microorganisms residing in the gastrointestinal tract may be a critical, previously overlooked biological modifier of radiotherapy response and toxicity. Gut microbiota may be an important player in modulating tumour microenvironment, ultimately affecting treatment efficacy. The gut microbiome can influence both the effectiveness of cancer treatment and the severity of cancer treatment induced gastrointestinal toxicities
Why should Medical Physicists care about the Gut Microbiome?
At first glance, the gut microbiome may appear far removed from beam modelling, inverse planning, or quality assurance. However, the microbiome directly influences several processes that are central to radiotherapy:
- Radiation-induced inflammation
- DNA damage response and repair
- Immune activation and suppression
- Normal tissue regeneration
For pelvic, abdominal, and even extra abdominal radiotherapy, these biological effects can significantly alter the therapeutic ratio, irrespective of how well the dose is optimized.
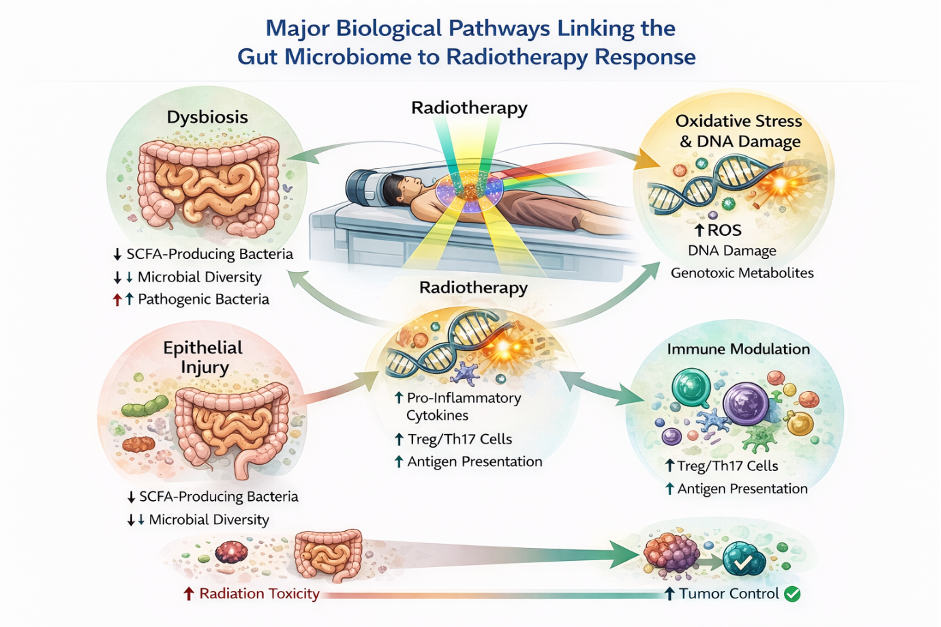
Radiotherapy induced Dysbiosis: An unintended Biological Effect
Ionizing radiation does not affect only tumour cells and normal tissues, it also alters gut microbial ecosystems. Radiotherapy can induce dysbiosis, characterized by:
- Reduced microbial diversity
- Loss of beneficial short-chain fatty acid (SCFA) producing bacteria
- Expansion of pro-inflammatory and pathogenic species
These changes are particularly pronounced during:
- Pelvic IMRT (cervical, prostate, rectal cancers)
- Abdominal SBRT
- Chemoradiation protocols
Importantly, dysbiosis may persist long after treatment, contributing to chronic radiation toxicity.
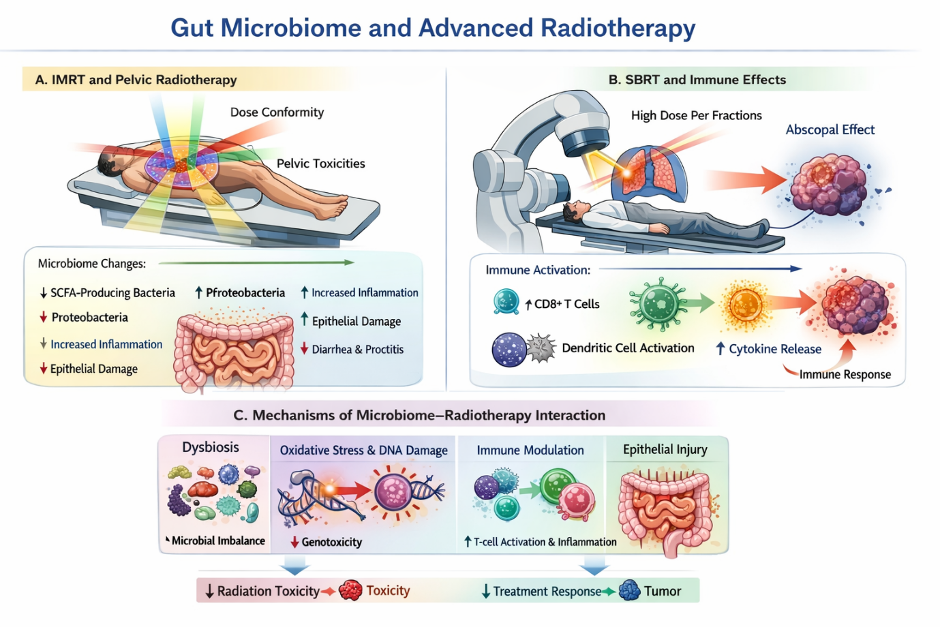
IMRT and the microbiome: more than bowel dose constraints
IMRT has significantly reduced high-dose exposure to bowel loops, yet acute diarrhoea and chronic radiation proctitis remain common. Recent clinical studies indicate that:
- Patients with low baseline gut microbial diversity experience higher grade gastrointestinal toxicity
- Loss of Faecalibacterium prausnitzii (a butyrate producing bacterium) correlates with mucosal inflammation
- Increased Proteobacteria abundance is associated with severe proctitis
This suggests that two patients with identical DVHs may respond very differently due to underlying microbiome differences, a paradigm familiar to physicists working in Radiogenomics, now extending into Radiomicrobiomics.
SBRT, High Dose per Fraction, and Immune Modulation
SBRT introduces unique radiobiological effects:
- High dose per fraction
- Vascular damage
- Strong immune activation
The gut microbiome modulates these effects by shaping systemic immune responses. Preclinical and early clinical evidence suggests that:
- Favourable microbiome profiles enhance immune mediated tumour control
- Antibiotic induced microbiome depletion reduces SBRT efficacy
- The microbiome influences the likelihood of observing the abscopal effect
For medical physicists involved in SBRT planning, this reinforces the idea that biological context matters as much as geometric accuracy.
Pelvic and abdominal radiotherapy: A Microbiome-critical site
Pelvic and abdominal radiotherapy represents the most clinically relevant interface between radiation and the gut microbiome. Radiation induced changes in gut flora contribute to:
| Clinical Effect | Microbiome Contribution |
| Acute diarrhoea | Inflammatory dysbiosis |
| Chronic proctitis | Persistent microbial imbalance |
| Late enteropathy | Impaired epithelial regeneration |
Increased toxicity burden may lead to treatment interruptions and therefore, from a workflow perspective, this highlights the importance of integrated supportive care, including dietary counselling and microbiome preserving strategies during treatment.
Radiotherapy induced dysbiosis amplifies oxidative stress, DNA damage, inflammatory cytokine release, and immune dysregulation. Altered antigen presentation and imbalance between regulatory (Treg) and effector (Th17/CD8⁺) immune cells influence both tumour control and normal tissue injury. These pathways operate independently of physical dose parameters and help explain inter-patient variability in outcomes.
The microbiome immune radiotherapy axis
With the deeper understanding of radiobiology underlying radiotherapy management of cancer, it is now recognized that radiotherapy works as an immune modulating treatment. The gut microbiome acts as a biological amplifier of this effect by:
- Enhancing antigen presentation
- Promoting CD8+ T-cell activation
- Regulating cytokine signalling
This triad is especially relevant in combined radiotherapy with immunotherapy protocols, where microbiome composition can determine treatment success or failure.
Clinical translation: what can be done today?
While microbiome guided radiotherapy planning is not yet routine, several practical considerations are already relevant:
- Avoid unnecessary antibiotics during radiotherapy
- Encourage high fibre diets to support SCFA production
- Consider probiotics cautiously in selected patients
- Recognize unexplained toxicity as a possible biological and not as dosimetric phenomenon
Future integration may include baseline microbiome profiling alongside imaging and dosimetric data.
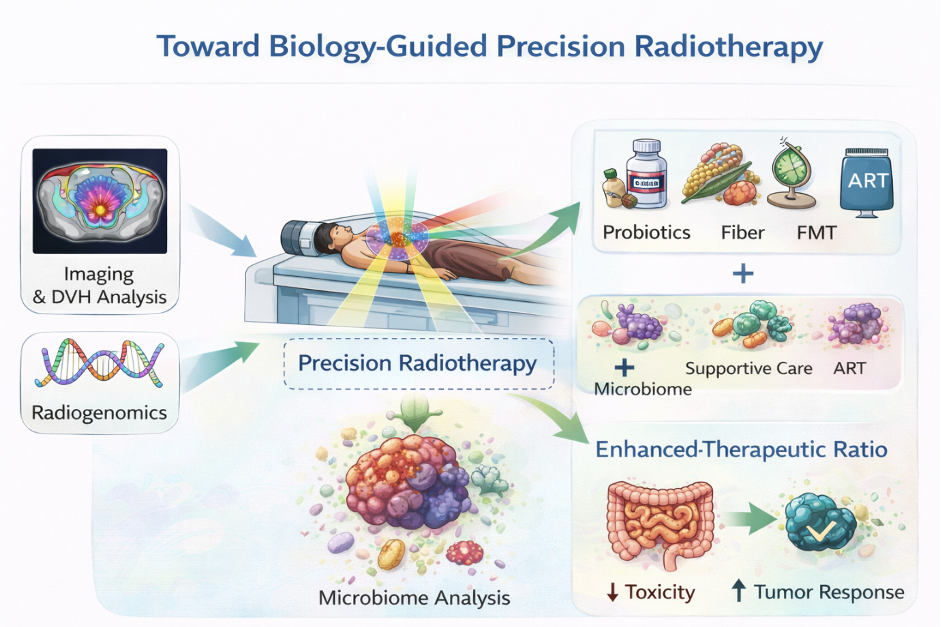
Implications of understanding of microbiome effect by Medical Physicists
The evolution of radiotherapy has progressed from conventional dose delivery to advanced technics such as CRT, IMRT, IGRT and the next frontier can be biology guided radiotherapy, where microbiome data complements Radiogenomics, functional imaging, and AI-driven planning. For medical physicists, this represents an opportunity to expand the scope of precision from shaping dose distributions to understanding biological variability.
Conclusion
The gut microbiome is emerging as a significant biological determinant of radiotherapy response and toxicity, particularly in IMRT, SBRT, and pelvic radiotherapy. While advanced technology has optimized physical dose delivery, biological heterogeneity continues to shape clinical outcomes. Integrating microbiome science into radiation oncology offers a promising pathway toward truly personalized radiotherapy where physics and biology work in harmony to improve patient care.
Further readings
- Jing Liu, Chao Liu, Jinbo Yue : Radiotherapy and the gut microbiome: facts and fiction. Radiat Oncol. 2021 Jan 13;16:9. doi: 10.1186/s13014-020-01735-9
- Gerassy-Vainberg, S., Blatt, A., Danin-Poleg, Y., Gershovich, K., Sabo, E., Nevelsky, A., Daniel, S., Dahan, A., Ziv, O., Dheer, R., Abreu, M. T., Koren, O., Kashi, Y., & Chowers, Y. (2018). Radiation induces proinflammatory dysbiosis: Transmission of inflammatory susceptibility by host cytokine induction. Gut, 67(1), 97-107. https://doi.org/10.1136/gutjnl-2017-313789
- Li-Wei Xie et al.: Gut microbiota and radiation-induced injury: mechanistic insights and microbial therapies. Gut Microbes. Volume 17, 2025 https://doi.org/10.1080/19490976.2025.2528429
- Beth A Helmink , M A Wadud Khan, Amanda Hermann, Vancheswaran Gopalakrishnan, Jennifer A Wargo :The microbiome, cancer, and cancer therapy. Nat Med. 2019 Mar;25(3):377-388. doi: 10.1038/s41591-019-0377-7.
